Currently, therapeutic drug monitoring approach, which involves the measurement of drug concentrations and their interpretation, has become a standard of care in immunosuppressant therapy for dose optimization, with the aim of maximizing therapeutic benefits and minimizing adverse effects. A non-monitored dosing can increase the risk for therapeutic failure or induce serious undesirable effects. Therapeutic drug monitoring is a common practice for the use of immunosuppressant drugs, which generally exhibit considerable inter- or intra- pharmacokinetic (PK) variability and narrow therapeutic window. Therefore, for B-LSS application, Pop-PK model diagnostic criteria should additionally account for AUC prediction errors. However, B-LSS performance is not perfectly in line with the standard Pop-PK model selection criteria hence the final model might not be ideal for AUC prediction purpose. Conclusionsī-LSS can adequately estimate cyclosporine AUC. Moreover, B-LSS perform better for the prediction of the ‘underlying’ AUC derived from the Pop-PK model estimated concentrations that exclude the residual errors, in comparison to their prediction of the observed AUC directly calculated using measured concentrations. Twelve B-LSS, consisting of 4 or less sampling points obtained within 4 hours post-dose, predict AUC with 95 th percentile of the absolute values of relative prediction errors of 20% or less. The best performing models for intravenous and oral cyclosporine are the structure ones with combined and additive error, respectively. The final covariate model does not improve the B-LSS prediction performance. ResultsĪ two-compartment structure model with a lag time and a combined additive and proportional error is retained. The performance of B-LSS when targeting different versions of AUC was also discussed. Pop-PK analyses were carried out and the predictive performance of B-LSS was evaluated using the final Pop-PK model and several related ones. Twenty five pediatric hematopoietic stem cell transplantation patients receiving intravenous and oral cyclosporine were investigated.
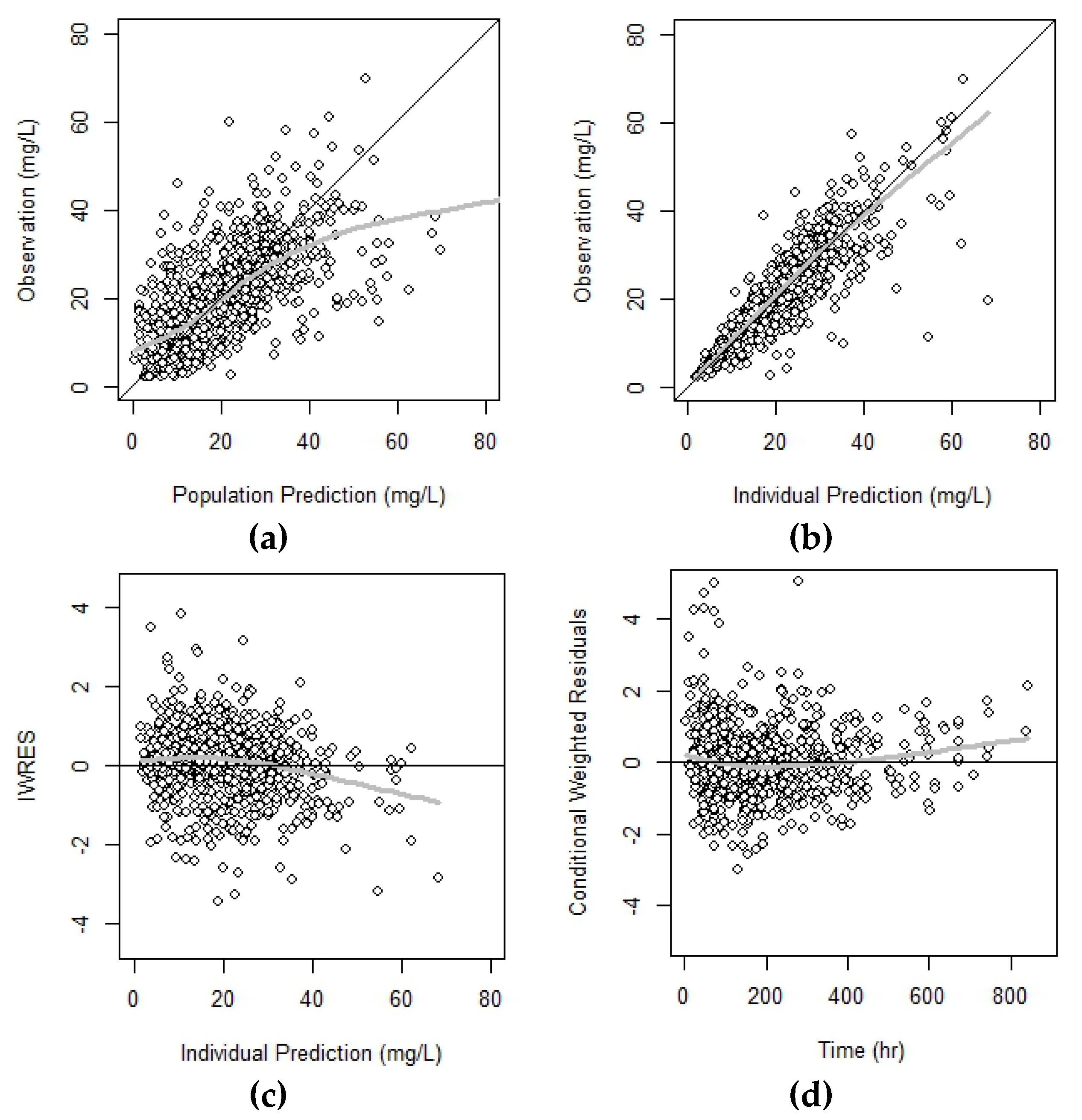
In this paper, we develop Bayesian limited sampling strategies (B-LSS) for cyclosporine AUC estimation using population pharmacokinetic (Pop-PK) models and investigate related issues, with the aim to improve B-LSS prediction performance. However, there is a growing interest in the use of the area under the concentration-time curve (AUC), particularly for cyclosporine dose adjustment in pediatric hematopoietic stem cell transplantation. The optimal marker for cyclosporine (CsA) monitoring in transplantation patients remains controversial.
